Lens implant
Surgeons annually implant more than 6 million lenses. The procedure can be done under local anesthesia with the patient awake throughout the operation. The use of a flexible IOL enables the lens to be rolled for insertion into the capsule through a very small incision, thus avoiding the need for stitches. This procedure usually takes less than 30 minutes in the hands of an experienced ophthalmologist. The recovery period is about 2–3 weeks. After surgery, patients should avoid strenuous exercise or anything else that significantly increases blood pressure. They should visit their ophthalmologists regularly for several months to monitor the implants.
IOL implantation carries several risks associated with eye surgeries, such as infection, loosening of the lens, lens rotation, inflammation and nighttime halos, but a systematic review of studies has determined that the procedure is safer than conventional laser eye treatment. Though IOLs enable many patients to have reduced dependence on glasses, most patients still rely on glasses for certain activities, such as reading.

Intraocular lenses have been used since 1999 for correcting larger errors in near-sighted, far-sighted, and astigmatic eyes. This type of IOL is also called phakic intraocular lens (PIOL), and the crystalline lens is not removed.
Phakic IOL appear to be less dangerous than excimer laser surgery in those with significant nearsightedness.
More commonly, aphakic IOLs (that is, not PIOLs) are implanted via clear lens extraction and replacement (CLEAR) surgery. During CLEAR, the crystalline lens is extracted and an IOL replaces it in a process that is very similar to cataract surgery: both involve lens replacement, local anesthesia, last approximately 30 minutes, and require making a small incision in the eye for lens insertion. People recover from CLEAR surgery 1–7 days after the operation. During this time, they should avoid strenuous exercise or anything else that significantly raises blood pressure. They should visit their ophthalmologists regularly for several months to monitor the IOL implants.
CLEAR has a 90% success rate (risks include wound leakage, infection, inflammation, and astigmatism). CLEAR can be performed only on patients ages 40 and older. This is to ensure that eye growth, which disrupts IOL lenses, will not occur post-surgery.
Once implanted, IOL lenses have three major benefits. First, they are an alternative to LASIK, a form of eye surgery that does not work for people with serious vision problems. Effective IOL implants also entirely eliminate the need for glasses or contact lenses post-surgery for most patients. The cataract will not return, as the lens has been removed. The disadvantage is that the eye’s ability to change focus (accommodate) has generally been reduced or eliminated, depending on the kind of lens implanted.
Some of the risks that FDA have been found so far during a three-year study of the Artisan are:
- a yearly loss of 1.8% of the endothelial cells,
- 0.6% risk of retinal detachment,
- 0.6% risk of cataract (other studies have shown a risk of 0.5 – 1.0%), and
- 0.4% risk of corneal swelling.
Other risks include:
- 0.03–0.05% eye infection risk, which in worst case can lead to blindness. This risk exists in all eye surgery procedures and is not unique for IOLs.
- glaucoma,
- astigmatism,
- remaining near or far sightedness,
- rotation of the lens inside the eye one or two days after surgery.
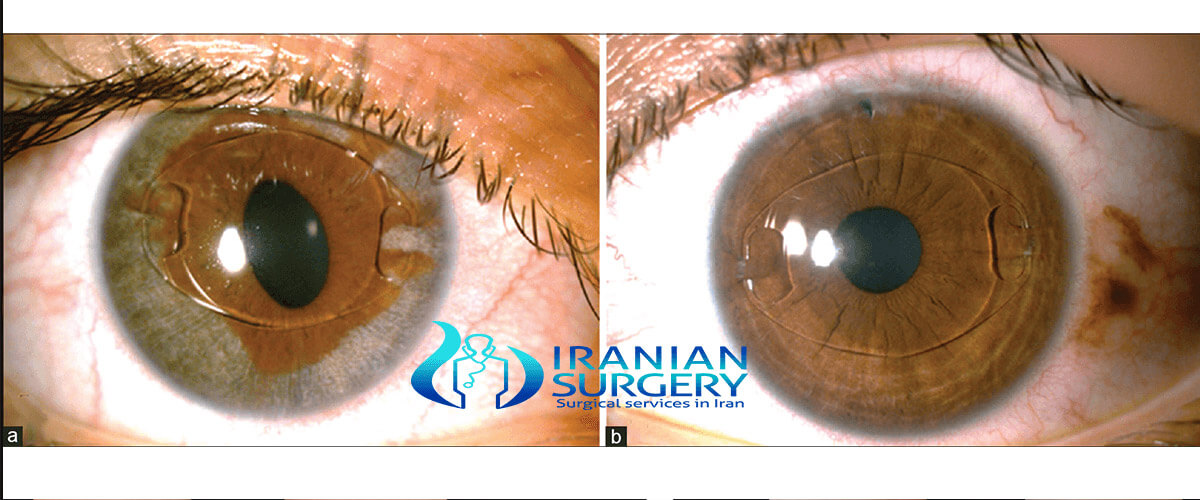
One of the causes of the risks above is that the lens can rotate inside the eye if the PIOL is too short, if the eye was incorrectly measured, or because the sulcus has a slightly oval shape (the height is slightly smaller than the width). Toric IOLs must be powered and aligned inside the eye on a meridian that corrects the patient’s preexisting astigmatism. Again, these lenses can rotate inside the eye postoperatively or be placed incorrectly by the operating surgeon. Either way, the patient’s preexisting astigmatism may not be corrected completely or may even increase.
When IOLs are implanted in the aphakic patient—either after clear lensectomy for the correction of refractive error or after cataract extraction to restore eyesight—astigmatism in these patients is mainly attributable to the cornea. The surgeon can ascertain the astigmatic, or steepest, meridian in a number of ways, including manifest refraction or corneal topography. Manifest refraction is the familiar test where the eye doctor rotates lenses in front of the eye, asking the patient, “Which is better (or clearer), this one or this one?” Corneal topography is considered a more quantitative test, and for purposes of aligning a toric IOL, most surgeons use a measurement called simulated keratometry (SimK), which is calculated by the internal programming of the corneal topography machine, to determine the astigmatic meridian on the surface of the cornea. The astigmatic meridian can also be identified using corneal wavefront technology or paraxial curvature matching.
Classifications of States of the Eye (Before, During, or After Cataract Extraction)
- Phakia is the presence of the natural crystalline lenses. Phakic IOLs (PIOLs) are implanted without removal of the patient’s original crystalline lens, and this is performed solely to correct refractive error in the presence of a clear crystalline lens.
- Aphakia is the absence of the natural crystalline lens. The aphakic state is usually due to surgery to remove a cataractous lens, but post-surgical aphakia is rare nowadays because of the ubiquity of intraocular lenses. Rarely, aphakia can be post-traumatic or congenital in nature. “Aphakic IOLs” generally refer to lenses implanted secondarily in an eye already aphakic from previous surgery or trauma some time ago.
- Pseudophakia is the substitution of the natural crystalline lens with an IOL, as is often done after cataract extraction or to correct refractive error. “Pseudophakic IOLs” refer to lenses implanted during cataract surgery, as a sequential step after removal of the cataractous lens of the person.
Many aphakic and pseudophakic IOLs such as anterior chamber IOLs or 3 piece posterior chamber IOLs can be used interchangeably. The exception are one piece IOLs, which must be placed within the capsular bag at the time of cataract surgery and hence cannot be used as secondary implants.
Types of Intraocular Lenses (IOLs)
Classified by Implanted Location
Anterior Chamber IOLs (ACIOLs)
A less-common type of intraocular lens, which is sometimes used if a PCIOL is not an option for a patient or if the situation requires a phakic IOL (PIOL).
Posterior Chamber IOLs (PCIOLs)
This is by far the most common type of implanted lens after cataract surgery in the United States.
Classified by Technical Specifications
Toric IOLs
A toric IOL is a type of toric lens used to correct preexisting corneal astigmatism at the time of cataract surgery. This astigmatism can also be treated with limbal relaxing incisions or an excimer laser procedure. About 40% of Americans have significant astigmatism and thus may be candidates for a toric IOL. Cataract surgery with implantation of a toric IOL is essentially the same as cataract surgery with a conventional IOL. Like toric contact lenses, toric IOLs have different powers in different meridians of the lens, and they must be positioned on the correct meridian to reverse the preexisting astigmatism. If the toric IOL is not on the correct meridian, it may need to be repositioned in a second procedure.
PIOLs
PIOLs intraocular lenses which are placed in an eye that still contains a natural human crystalline lens. PIOLs are sometimes referred to as an ‘implantable contact lenses’ (ICLs). As with other IOLs, phakic IOLs (PIOLs) can be spheric or toric. Toric PIOLs have to be aligned with the meridian of astigmatism; toric IOL misalignment or rotation can lead to residual or even greater astigmatism postoperatively.
Depending on their attachment site to the eye, PIOLs can be divided into:
- Angle-supported PIOLs: those IOLs are placed in the anterior chamber. They are notorious for their negative impact on the corneal endothelial lining, which is vital for maintaining a healthy clear cornea.
- Iris-supported PIOLs: The IOL is attached by claws to the mid-peripheral iris by a technique called enclavation. It is believed to have a lesser effect on corneal endothelium. The main complication with this type is their tendency to cause endothelial cell reduction.
- Sulcus-supported PIOLs: this type is gaining more and more popularity. These IOLs are placed in the posterior chamber in front of the natural crystalline lens. They have special vaulting so as not to be in contact with the normal lens. The main complication with older versions was a small possibility of cataract formation.
Pseudophakic IOLs That Address Presbyopia
One of the major disadvantages of conventional IOLs is that they are primarily focused for distance vision. Though patients who undergo a standard IOL implantation no longer experience clouding from cataracts, they are unable to accommodate, or change focus from near to far, far to near, and to distances in between. In the United States, a new category of intraocular lenses was opened with the approval by the Food and Drug Administration in 2003 of multifocal and accommodating lenses. These come at an additional cost to the recipient beyond what Medicare will pay and each has advantages and disadvantages.
Monofocal lenses are standard lenses used in cataract surgery. People who have a multifocal intraocular lens after their cataract is removed may be less likely to need additional glasses compared with people who have standard monofocal lenses. However, people receiving multifocal lenses may experience more visual problems, such as glare or haloes (rings around lights), than with monofocal lenses.
Multifocal IOLs
Multifocal IOLs attempt to provide simultaneous viewing of distance vision and near vision. Trifocal IOLs can provide intermediate vision. The most common adverse visual effects from multifocal IOLs include glare, halos, and a loss of contrast sensitivity, in low light conditions. Many multifocal IOL designs attempt to achieve this simultaneous viewing focus using a concentric ring design, which alternates distance and near focal points. However, many concentric ring multifocal lenses used today are prone to glare and mildly compromised focus at all ranges of vision.
Accommodating IOLs
Some newer lens designs attempt to allow the eye to regain some partial focusing ability in order to change focus from distance to near (accommodation). However, many accommodating IOLs used today only achieve a very limited improvements in near vision which reduced over time. Accommodative intraocular lenses may also have a slightly higher risk of developing posterior capsule opacification (PCO), though there is some uncertainty around this finding. PCO is a common side-effect of many cataract surgeries and is easily treatable with a one-time laser capsulotomy procedure.
Accommodating IOLs interact with ciliary muscles and zonules, using hinges at both ends to “latch on” and move forward and backward inside the eye using the same mechanism as normal accommodation. These IOLs have a 4.5-mm square-edged optic and a long hinged plate design with polyimide loops at the end of the haptics. The hinges are made of an advanced silicone called BioSil that was thoroughly tested to make sure it was capable of unlimited flexing in the eye.
Posterior Capsular Opacities (“After Cataracts”)
A small percentage of posterior chamber intraocular lenses (PCIOLs) may form posterior capsular opacities (PCO), commonly referred to as “after cataracts.” Posterior capsular opacities are easily treatable, and typically only require a one-time capsulotomy procedure (using a Nd:YAG laser) to clarify.
Materials
The materials that have been used to manufacture intraocular lens implants include polymethylmethacrylate (PMMA), silicone, hydrophobic acrylate, hydrophilic acrylate and collamer. Polymethylmethacrylate (PMMA) was the first material to be used successfully in intraocular lenses. British ophthalmologist Sir Harold Ridleyobserved that Royal Air Force pilots who sustained eye injuries during World War II involving PMMA windshield material did not show any rejection or foreign bodyreaction. Deducing that the transparent material was inert and useful for implantation in the eye, Ridley designed and implanted the first intraocular lens in a human eye.
Advances in technology have brought about the use of silicone and acrylic, both of which are soft foldable inert materials. This allows the lens to be folded and inserted into the eye through a smaller incision. Specifically, acrylic lenses are a better choice in people who have a history of uveitis or are likely to have to undergo retinal surgery requiring vitrectomy with replacement by silicone oil, such as persons with proliferative diabetic retinopathy or who are at high risk of retinal detachment, such as persons with high myopia. A study found that in participants with a history of uveitis, eyes treated with hydrophobic acrylic IOLs were over 2 times more likely to have a best corrected visual acuity of 20/40 or more, compared to eyes treated with silicone IOLs.
Blue light filtering IOLs filter the UV and high-energy blue light present in natural and artificial light, both of which can cause vision problems; however too much filtering of blue light can increase depression, especially in the winter months (SAD). The trademarked “Natural Yellow” material is available in three hydrophilic IOLs. Dr. Patrick H. Benz of Benz Research and Development created the first IOL material to incorporate the same UV-A blocking and violet light filtering chromophore that’s present in the human crystalline lens in order to attempt to protect the retina after cataract extraction of the natural crystalline lens.
References
- ^ Güell, Jose Luis; Morral, Merce; Kook, Daniel; Kohnen, Thomas (2010). “Phakic intraocular lenses”. Journal of Cataract & Refractive Surgery. 36 (11): 1976–1993. doi:10.1016/j.jcrs.2010.08.014. PMID 21029908.
- ^ Sanders, Donald; Vukich, John A (2006). “Comparison of Implantable Collamer Lens (ICL) and Laser-assisted in Situ Keratomileusis (LASIK) for Low Myopia”. Cornea. 25 (10): 1139–46. doi:10.1097/ICO.0b013e31802cbf3c. PMID 17172886.
- ^ Gaudet, Jodie ed. 1001 Inventions That Changed the World, page 697, ISBN 978-0-7333-2536-6
- https://en.wikipedia.org/wiki/Intraocular_lens
المرجع:
[kkstarratings]