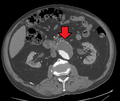
Abdominal aortic aneurysm
Abdominal aortic aneurysm (AAA or triple A) is a localized enlargement of the abdominal aorta such that the diameter is greater than 3 cm or more than 50% larger than normal. They usually cause no symptoms, except during rupture.Occasionally, abdominal, back, or leg pain may occur. Large aneurysms can sometimes be felt by pushing on the abdomen. Rupture may result in pain in the abdomen or back, low blood pressure, or loss of consciousness, and often results in death.
AAAs occur most commonly in those over 50 years old, in men, and among those with a family history. Additional risk factors include smoking, high blood pressure, and other heart or blood vessel diseases. Genetic conditions with an increased risk include Marfan syndrome and Ehlers-Danlos syndrome.AAAs are the most common form of aortic aneurysm. About 85% occur below the kidneys with the rest either at the level of or above the kidneys. In the United States, screening with abdominal ultrasound is recommended for males between 65 and 75 years of age with a history of smoking. In the United Kingdom and Sweden, screening all men over 65 is recommended. Once an aneurysm is found, further ultrasounds are typically done on a regular basis.
Not smoking is the single best way to prevent the disease. Other methods of prevention include treating high blood pressure, treating high blood cholesterol, and not being overweight. Surgery is usually recommended when the diameter of an AAA grows to >5.5 cm in males and >5.0 cm in females. Other reasons for repair include the presence of symptoms and a rapid increase in size, defined as more than one centimeter per year. Repair may be either by open surgery or endovascular aneurysm repair (EVAR). As compared to open surgery, EVAR has a lower risk of death in the short term and a shorter hospital stay, but may not always be an option. There does not appear to be a difference in longer term outcomes between the two.Repeat procedures are more common with EVAR.
AAAs affect 2-8% of males over the age of 65. Rates among women are one-fourth as high. In those with an aneurysm less than 5.5 cm, the risk of rupture in the next year is below 1%. Among those with an aneurysm between 5.5 and 7 cm, the risk is about 10%, while for those with an aneurysm greater than 7 cm the risk is about 33%. Mortality if ruptured is 85% to 90%. During 2013, aortic aneurysms resulted in 168,200 deaths, up from 100,000 in 1990. In the United States AAAs resulted in between 10,000 and 18,000 deaths in 2009.

Signs and symptoms
The vast majority of aneurysms are asymptomatic. However, as abdominal aortic aneurysms expand, they may become painful and lead to pulsating sensations in the abdomen or pain in the chest, lower back, or scrotum. The risk of rupture is high in a symptomatic aneurysm, which is therefore considered an indication for surgery. The complications include rupture, peripheral embolization, acute aortic occlusion, and aortocaval (between the aorta and inferior vena cava) or aortoduodenal (between the aorta and the duodenum) fistulae. On physical examination, a palpable and pulsatile abdominal mass can be noted. Bruits can be present in case of renal or visceral arterial stenosis.
Aortic rupture
The signs and symptoms of a ruptured AAA may include severe pain in the lower back, flank, abdomen or groin. A mass that pulses with the heart beat may also be felt. The bleeding can lead to a hypovolemic shock with low blood pressure and a fast heart rate. This may lead to brief passing out.
The mortality of AAA rupture is as high as 90 percent. 65 to 75 percent of patients die before they arrive at the hospital and up to 90 percent die before they reach the operating room. The bleeding can be retroperitoneal or into the abdominal cavity. Rupture can also create a connection between the aorta and intestine or inferior vena cava. Flank ecchymosis (appearance of a bruise) is a sign of retroperitoneal bleeding, and is also called Grey Turner’s sign.
Aortic aneurysm rupture may be mistaken for the pain of kidney stones, or muscle related back pain
Causes
The exact causes of the degenerative process remain unclear. There are, however, some hypotheses and well-defined risk factors.
- Tobacco smoking: More than 90% of people who develop an AAA have smoked at some point in their lives.
- Alcohol and hypertension: The inflammation caused by prolonged use of alcohol and hypertensive effects from abdominal edema which leads to hemorrhoids, esophageal varices, and other conditions, is also considered a long-term cause of AAA.
- Genetic influences: The influence of genetic factors is high. AAA is four to six times more common in male siblings of known patients, with a risk of 20-30%. The high familial prevalence rate is most notable in male individuals. There are many hypotheses about the exact genetic disorder that could cause higher incidence of AAA among male members of the affected families. Some presumed that the influence of alpha 1-antitrypsin deficiency could be crucial, while other experimental works favored the hypothesis of X-linked mutation, which would explain the lower incidence in heterozygous females. Other hypotheses of genetic causes have also been formulated. Connective tissue disorders, such as Marfan syndrome and Ehlers-Danlos syndrome, have also been strongly associated with AAA.Both relapsing polychondritis and pseudoxanthoma elasticum may cause abdominal aortic aneurysm.
- Atherosclerosis: The AAA was long considered to be caused by atherosclerosis, because the walls of the AAA frequently carry an atherosclerotic burden. However, this hypothesis cannot be used to explain the initial defect and the development of occlusion, which is observed in the process.
- Other causes of the development of AAA include: infection, trauma, arteritis, and cystic medial necrosis.
Pathophysiology
The most striking histopathological changes of the aneurysmatic aorta are seen in the tunica media and intima layers. These changes include the accumulation of lipids in foam cells, extracellular free cholesterol crystals, calcifications, thrombosis, and ulcerations and ruptures of the layers. Adventitial inflammatory infiltrate is present.However, the degradation of the tunica media by means of a proteolytic process seems to be the basic pathophysiologic mechanism of AAA development. Some researchers report increased expression and activity of matrix metalloproteinases in individuals with AAA. This leads to elimination of elastin from the media, rendering the aortic wall more susceptible to the influence of blood pressure. Other reports have suggested the serine protease granzyme B may contribute to aortic aneurysm rupture through the cleavage of decorin, leading to disrupted collagen organization and reduced tensile strength of the adventitia. There is also a reduced amount of vasa vasorum in the abdominal aorta (compared to the thoracic aorta); consequently, the tunica media must rely mostly on diffusion for nutrition, which makes it more susceptible to damage.
Hemodynamics affect the development of AAA, which has a predilection for the infrarenal aorta. The histological structure and mechanical characteristics of the infrarenal aorta differ from those of the thoracic aorta. The diameter decreases from the root to the aortic bifurcation, and the wall of the infrarenal aorta also contains a lesser proportion of elastin. The mechanical tension in the abdominal aortic wall is therefore higher than in the thoracic aortic wall. The elasticity and distensibility also decline with age, which can result in gradual dilatation of the segment. Higher intraluminal pressure in patients with arterial hypertension markedly contributes to the progression of the pathological process. Suitable hemodynamic conditions may be linked to specific intraluminal thrombus (ILT) patterns along the aortic lumen, which in turn may affect AAA’s development.
Diagnosis
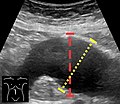
An abdominal aortic aneurysm is usually diagnosed by physical exam, abdominal ultrasound, or CT scan. Plain abdominal radiographs may show the outline of an aneurysm when its walls are calcified. However, this is the case in less than half of all aneurysms. Ultrasonography is used to screen for aneurysms and to determine the size of any present. Additionally, free peritoneal fluid can be detected. It is noninvasive and sensitive, but the presence of bowel gas or obesity may limit its usefulness. CT scan has a nearly 100% sensitivity for an aneurysm and is also useful in preoperative planning, detailing the anatomy and possibility for endovascular repair. In the case of suspected rupture, it can also reliably detect retroperitoneal fluid. Alternative less often used methods for visualization of an aneurysm include MRIand angiography.
An aneurysm ruptures if the mechanical stress (tension per area) exceeds the local wall strength; consequently, peak wall stress (PWS) and peak wall rupture risk (PWRR) have been found to be more reliable parameters than diameter to assess AAA rupture risk. Medical software allows computing these rupture risk indices from standard clinical CT data and provides a patient-specific AAA rupture risk diagnosis. This type of biomechanical approach has been shown to accurately predict the location of AAA rupture.
-
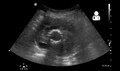
Ultrasound showing a previously repaired AAA that is leaking with flow around the graft
Classification
| Ectatic or mild dilatation |
>2.0 cm and <3.0 cm |
| Moderate | 3.0 – 5.0 cm |
| Large or severe | >5.0 or 5.5 cm |
Abdominal aortic aneurysms are commonly divided according to their size and symptomatology. An aneurysm is usually defined as an outer aortic diameter over 3 cm (normal diameter of the aorta is around 2 cm), or more than 50% of normal diameter. If the outer diameter exceeds 5.5 cm, the aneurysm is considered to be large. A ruptured AAA is a clinical diagnosis involving the presence of the triad of abdominal pain, shock, and a pulsatile abdominal mass. If these conditions are present, indicating AAA rupture, no further clinical investigations are needed before surgery.
The suprarenal aorta normally measures about 0.5 cm larger than the infrarenal aorta.
Prevention
- Smoking cessation
- Treatment of hypertension
Screening
The U.S. Preventive Services Task Force (USPSTF) recommends a single screening abdominal ultrasound for abdominal aortic aneurysm in males age 65 to 75 years who have a history of smoking. It is unclear if screening is useful in women who have smoked and the USPSTF recommend against screening in women who have never smoked.
In the United Kingdom the NHS AAA Screening Programme invites men in England for screening during the year they turn 65. Men over 65 can contact the programme to arrange to be screened.
In Sweden one time screening is recommended in all males over 65 years of age. This has been found to decrease the risk of death from AAA by 42% with a number needed to screen of just over 200.In those with a close relative diagnosed with an aortic aneurysm, Swedish guidelines recommend an ultrasound at around 60 years of age.
Australia has no guideline on screening.
Repeat ultrasounds should be carried out in those who have an aortic size greater than 3.0 cm. In those whose aorta is between 3.0 and 3.9 cm this should be every three years, if between 4.0 and 4.4 cm every two years, and if between 4.5 and 5.4 cm every year.
Management
The treatment options for asymptomatic AAA are conservative management, surveillance with a view to eventual repair, and immediate repair. Two modes of repair are available for an AAA: open aneurysm repair, and endovascular aneurysm repair (EVAR). An intervention is often recommended if the aneurysm grows more than 1 cm per year or it is bigger than 5.5 cm. Repair is also indicated for symptomatic aneurysms.
Conservative
Conservative management is indicated in people where repair carries a high risk of mortality and in patients where repair is unlikely to improve life expectancy. The mainstay of the conservative treatment is smoking cessation.
Surveillance is indicated in small asymptomatic aneurysms (less than 5.5 cm) where the risk of repair exceeds the risk of rupture. As an AAA grows in diameter, the risk of rupture increases. Surveillance until an aneurysm has reached a diameter of 5.5 cm has not been shown to have a higher risk as compared to early intervention.
Medication
No medical therapy has been found to be effective at decreasing the growth rate or rupture rate of asymptomatic AAAs.Blood pressure and lipids should, however, be treated per usual.
Surgery
The threshold for repair varies slightly from individual to individual, depending on the balance of risks and benefits when considering repair versus ongoing surveillance. The size of an individual’s native aorta may influence this, along with the presence of comorbidities that increase operative risk or decrease life expectancy. Evidence, however, does not support repair if the size is between 4 cm and 5.5 cm.
Open repair
Open repair is indicated in young patients as an elective procedure, or in growing or large, symptomatic or ruptured aneurysms. The aorta must be clamped off during the repair, denying blood to the abdominal organs and sections of the spinal cord; this can cause a range of complications. It is essential to make the critical part of the operation fast, so the incision is typically made large enough to facilitate the fastest repair. Recovery after open AAA surgery takes significant time. The minimums are a few days in intensive care, a week total in the hospital and a few months before full recovery.
Endovascular repair
Endovascular repair first became practical in the 1990s and although it is now an established alternative to open repair, its role is yet to be clearly defined. It is generally indicated in older, high-risk patients or patients unfit for open repair. However, endovascular repair is feasible for only a proportion of AAAs, depending on the morphology of the aneurysm. The main advantages over open repair are that there is less peri-operative mortality, less time in intensive care, less time in hospital overall and earlier return to normal activity. Disadvantages of endovascular repair include a requirement for more frequent ongoing hospital reviews, and a higher chance of further procedures being required. According to the latest studies, the EVAR procedure does not offer any benefit for overall survival or health-related quality of lifecompared to open surgery, although aneurysm-related mortality is lower. In patients unfit for open repair, EVAR plus conservative management was associated with no benefit, more complications, subsequent procedures and higher costs compared to conservative management alone. Endovascular treatment for paraanastomotic aneurysms after aortobiiliac reconstruction is also a possibility. A 2017 Cochrane review found tentative evidence of no difference in outcomes between endovascular and open repair of ruptured AAA in the first month.
Rupture
In those with aortic rupture of the AAA, treatment is immediate surgical repair. There appears to be benefits to allowing permissive hypotension and limiting the use of intravenous fluids during transport to the operating room.
Prognosis
| AAA Size (cm) | Growth rate (cm/yr) | Annual rupture risk (%) |
|---|---|---|
| 3.0-3.9 | 0.39 | 0 |
| 4.0-4.9 | 0.36 | 0.5-5 |
| 5.0-5.9 | 0.43 | 3-15 |
| 6.0-6.9 | 0.64 | 10-20 |
| >=7.0 | – | 20-50 |
Although the current standard of determining rupture risk is based on maximum diameter, it is known that smaller AAAs that fall below this threshold (diameter<5.5 cm) may also rupture, and larger AAAs (diameter>5.5 cm) may remain stable. In one report, it was shown that 10–24% of ruptured AAAs were less than 5 cm in diameter. It has also been reported that of 473 non-repaired AAAs examined from autopsy reports, there were 118 cases of rupture, 13% of which were less than 5 cm in diameter. This study also showed that 60% of the AAAs greater than 5 cm (including 54% of those AAAs between 7.1 and 10 cm) never experienced rupture. Vorp et al. later deduced from the findings of Darling et al. that if the maximum diameter criterion were followed for the 473 subjects, only 7% (34/473) of cases would have succumbed to rupture prior to surgical intervention as the diameter was less than 5 cm, with 25% (116/473) of cases possibly undergoing unnecessary surgery since these AAAs may never have ruptured.
Alternative methods of rupture assessment have been recently reported. The majority of these approaches involve the numerical analysis of AAAs using the common engineering technique of the finite element method (FEM) to determine the wall stress distributions. Recent reports have shown that these stress distributions have been shown to correlate to the overall geometry of the AAA rather than solely to the maximum diameter. It is also known that wall stress alone does not completely govern failure as an AAA will usually rupture when the wall stress exceeds the wall strength. In light of this, rupture assessment may be more accurate if both the patient-specific wall stress is coupled together with patient-specific wall strength. A non-invasive method of determining patient-dependent wall strength was recently reported, with more traditional approaches to strength determination via tensile testing performed by other researchers in the field. Some of the more recently proposed AAA rupture-risk assessment methods include: AAA wall stress; AAA expansion rate; degree of asymmetry; presence of intraluminal thrombus (ILT); a rupture potential index (RPI); a finite element analysis rupture index (FEARI); biomechanical factors coupled with computer analysis; growth of ILT; geometrical parameters of the AAA; and also a method of determining AAA growth and rupture based on mathematical models.
The post-operative mortality for an already ruptured AAA has slowly decreased over several decades but remains higher than 40%. However, if the AAA is surgically repaired before rupture, the post-operative mortality rate is substantially lower: approximately 1-6%.
Epidemiology
The occurrence of AAA varies by ethnicity. In the United Kingdom the rate of AAA in Caucasian men older than 65 years is about 4.7%, while in Asian men it is 0.45%. It is also less common in individuals of African, and Hispanic heritage. They occur four times more often in men than women.
There are at least 13,000 deaths yearly in the U.S. secondary to AAA rupture. The peak number of new cases per year among males is around 70 years of age, the percentage of males affected over 60 years is 2–6%. The frequency is much higher in smokers than in non-smokers (8:1), and the risk decreases slowly after smoking cessation. In the U.S., the incidence of AAA is 2–4% in the adult population.
Rupture of the AAA occurs in 1–3% of men aged 65 or more, the mortality is 70–95%.
References
- ^ Jump up to:Kent KC (27 November 2014). “Clinical practice. Abdominal aortic aneurysms”. The New England Journal of Medicine. 371 (22): 2101–8. doi:10.1056/NEJMcp1401430. PMID 25427112.
- ^ Jump up to:Upchurch GR, Schaub TA (2006). “Abdominal aortic aneurysm”. Am Fam Physician. 73 (7): 1198–204.
- https://en.wikipedia.org/wiki/Abdominal_aortic_aneurysm
[kkstarratings]